Fiber Coatings
Quick Start
If you coat a fibre with a liquid which can dry or cure, you might think that you would end up with a nice coated fibre. Instead you end up with a string of pearls, just like drops on a spider's web.
For those who happen not to know about the Rayleigh instability, this is very confusing. The beads appear at a specific spacing after a time t that depends on the radius r of the fibre, the coating thickness h and viscosity η. Times vary from μs to seconds because t depends on r4 and h3, so a pearl-free coating one day might be a string the next if a small change is made to the process.
Fiber Coatings
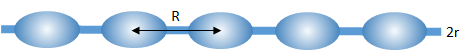 Coating onto a fiber doesn't seem all that hard. But it is commonly observed that the coating degenerates into a "string of pearls", with little droplets rather than a smooth coating.
Coating onto a fiber doesn't seem all that hard. But it is commonly observed that the coating degenerates into a "string of pearls", with little droplets rather than a smooth coating.
It turns out this is the Rayleigh instability. Drops on a fiber of radius r form with a spacing R given by:
`R = 2πsqrt(2)r`
The time, t, take for the instability to form depends also on the thickness of the coating, h, the viscosity η and surface tension σ and is given by:
`t = (12ηr^4)/(σh^3)`


